CNSystems Medizintechnik GmbH receives FDA Clearance
for Task Force® CORE
Graz, Austria, 13 April 2023 – CNSystems Medizintechnik GmbH is happy to announce that the U.S. Food and Drug Administration has cleared the 510(k) notification for CNSystem’s Task Force® CORE, permitting the sale of this product in the United States.

The Task Force® CORE measures continuous blood pressure and hemodynamics completely non-invasively and in real-time and is the state-of-the-art successor of the former Task Force® Monitor generation. Whenever continuous cardiovascular assessment is necessary, intermittent tools are insufficient and continuous invasive methods are not indicated, the Task Force® CORE is the device of choice.
The clinically validated CNAP® finger sensor technology implemented in the Task Force® CORE enables an easy, efficient and comprehensive analysis of the cardio-vascular system for state‐of‐the‐art diagnostic assessment of syncope and autonomic function or any other application requiring enhanced hemodynamic information. Providing cost‐efficient first‐class medical treatment closing the cycle of research, prevention, diagnosis and therapy control is the goal.
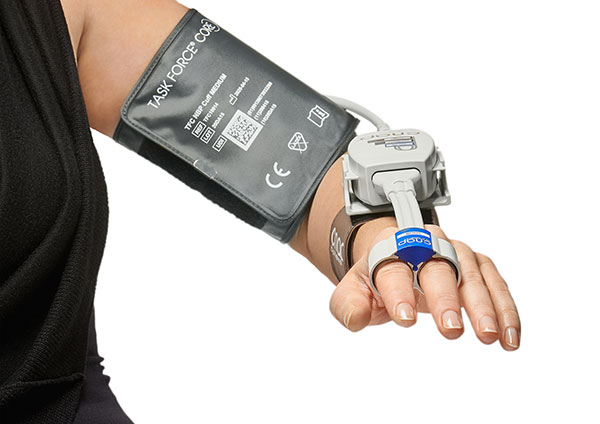
“The new Task Force® generation is already well received on the European market,” comments Dr. Jürgen Fortin, CEO of CNSystems, “and we are confident that the same will be true in the USA in cooperation with our dedicated partners on site.”
About CNSystems Medizintechnik GmbH:
Having started as a venture capital financed medical start‐up company, CNSystems has successfully supplied the world market with its non‐invasive, continuous hemodynamic diagnosis‐, research‐ and monitoring devices for 25 years.
The unique non‐invasive CNAP® finger sensor method increases patient outcome through continuous monitoring of blood pressure, cardiac output and further essential markers especially in areas where risky invasive measurements are not indicated.
Further information on CNSystems can be found at www.cnsystems.com.

